Hepatitis B is a common, serious viral infection that results in long-term smoldering liver inflammation, cirrhosis, and liver cancer that produce disability and early death. The CDC currently estimates 1.85 million US citizens suffer with chronic hepatitis B and that there are over 17,000 new cases annually, mainly in people aged 30-60, despite the availability of an effective vaccine since 1982.
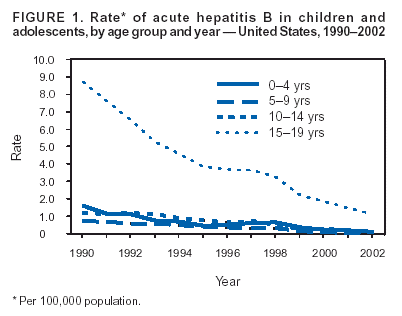
However, among young children we now have only 13-27 new cases annually (that is correct, one to two dozen, CDC data from 2015-1020), whereas before 1991 childhood hepatitis B cases consistently numbered 20,000 or more annually even with a vaccine available for 10 years.
What Happened 35 Years Ago to Make the Difference?
In late 1991 the CDC looked back at the poor results with hepatitis B since 1982 and concluded their approach to vaccinate selectively only infants of mothers who tested positive was a failure. There were simply too many sources of hepatitis B infection in early life (undiagnosed mothers, family members, asymptomatic friends). Universal vaccination was the right and essential approach — every infant, at birth, getting the hepatitis B vaccine and hepatitis B immune globulin. Then there was no window for infant infection.
That is the approach that has proven so successful and promptly led to a precipitous 99% drop in infantile cases of hepatitis B from 20,000 annually to a few handfuls by 2002. That low level has continued to this day, using several effective vaccines with excellent safety profiles (e.g., Engerix B and Recombivax).
What Just Happened in CDC?
In June, Robert F. Kennedy, Jr. (RFKJr), the attorney who generally opposes vaccines and who is secretary of the Department of Health and Human Services, dismissed all the vaccine experts who constituted the CDC’s Advisory Committee on Immunization Practices (ACIP) and replaced them with an assortment of people clearly lacking those qualifications.
That new ACIP this month revoked its 35-year-old recommendation for universal administration of hepatitis B vaccine at birth. The new CDC guidance is to screen birthing mothers for hepatitis B virus (HBV) and only administer the vaccine if the result is positive; otherwise, mothers who test negative are supposed to confer with their doctors to determine if they want their newborn to have the HBV vaccine at all, but still to delay the administration until age 2 months.
Back to the Future, Not in a Good Way
Note that this “new” protocol is almost the same as that used 1982-1991 when hepatitis B cases among newborns averaged 20,000 annually and were not reduced by the availability of vaccine, only worse because it adds a 2-month delay to the administration of the vaccine for no demonstrated reason. That delay represents a substantial and unnecessary window for infection to occur before the vaccine can provide protection.
What Can We Expect? MAHA or MAHHBA?
Unless birthing mothers and their physicians actually discuss the hepatitis B vaccine and overwhelmingly agree to give it to their infants while still in hospital, as now, we will have a major backsliding in protection. We know the baseline of 20,000 cases per year in infants from 35 years ago. We may not get there immediately, but the handfuls of cases as we have now will promptly become a thing of the past.
We will be rerunning a movie that we have seen before, repeating the documented experience from the 1980’s, with immense harm to come to thousands of infants who will not be protected from hepatitis B and who will become infected and victims of severe chronic liver disease at young ages.
RJKJr is provably wrong. This is not MAHA (Make America Healthy Again). This is MAHHBA (Make America Have Hepatitis B Again) and so, so wrong.